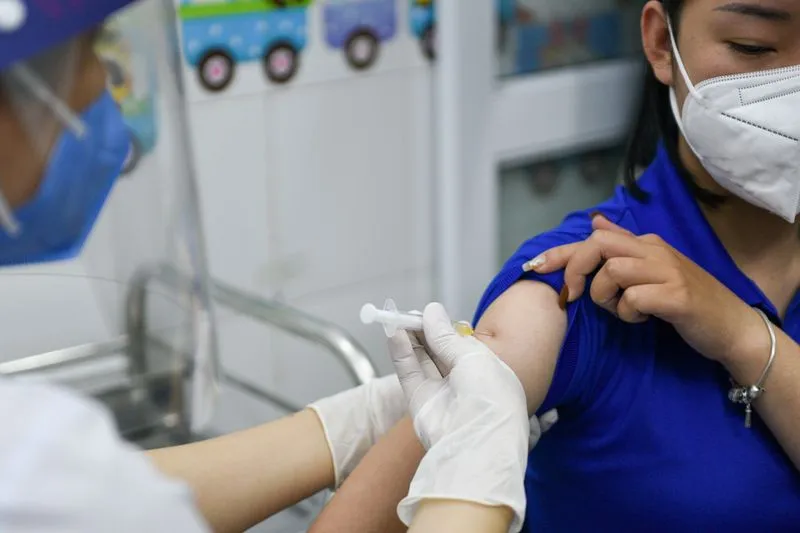
After reining in the virus for much of the pandemic, Vietnam faces a surge in daily infections. It has imposed strict curbs on movement in about a third of the nation, including the commercial hub of Ho Chi Minh City and the capital, Hanoi.
Local production of the unidentified mRNA vaccine could begin in the fourth quarter or early in 2022, foreign ministry spokeswoman Le Thi Thu Hang told a regular news briefing, adding that Vietnam could produce 100 million to 200 million doses a year under such a deal.
U.S. drug firms Pfizer and Moderna, which use new messenger RNA (mRNA) technology in their vaccines, did not immediately reply to Reuters’ requests for comments.
Such vaccines contain no actual virus, instead providing instructions for human cells to make proteins that mimic part of the coronavirus. These instructions spur the immune system into action, turning the body into a virus-zapping vaccine factory.
The health ministry reported 6,194 new COVID-19 infections on Thursday, a record daily increase. This raised Vietnam’s overall total of cases to 74,371, with 370 deaths.
Many Asian governments are looking to build up vaccine production at home as tight global supplies have hobbled their inoculation campaigns, which lag North America and Europe.
BioNTech has a production deal with China through local partner Fosun Pharma and plans to build a vaccine factory in Singapore that will begin operation in 2023. In South Korea, Samsung BioLogics plans to start vaccine bottling and packaging work for Moderna in the third quarter and the country is in talks with mRNA vaccine makers to make up to 1 billion doses.
Meanwhile the Vietnamese government said late on Thursday it was considering approving home-grown vaccine Nano Covax, the most promising out of four candidates, for emergency use soon.
The Nano Covax vaccine, based on recombinant DNA/protein technology, has been in third phase human trials on 13,000 volunteers since June 11 and is expected to be widely administered by end-2021, the government said in a statement.
PRODUCTION IN VIETNAM
In May, the World Health Organization said it was reviewing a proposal by an unidentified manufacturer in Vietnam to become an mRNA-based COVID-19 vaccine technology hub.
On Wednesday, Russia’s RDIF fund and Vietnamese firm Vabiotech said the latter had produced the first test batch of Sputnik V vaccine and was about to ship it to Russia for quality control checks.
Hang said Vabiotech would initially start packaging five million Sputnik V doses a month, using materials from Russia, and moving to produce 100 million doses a year later.
“Vabiotech is also in talks with a Japanese partner to soon transfer vaccine production technologies to Vietnam,” she added.
Vietnam’s domestic inoculation campaign is still at an early stage, with than 330,000 people have been fully vaccinated, official data showed.
The country will receive an additional 3 million shots of Moderna’s vaccine on July 25 from the United States via the global COVAX vaccine scheme, a Vietnamese foreign ministry spokeswoman said, adding to the 2 million Moderna doses the United States shipped it on July 10.
In total Vietnam – with a population of 98 million – has clinched deals for 105 million vaccine doses and is in talks for 70 million more.